Female Sexual Interest and Arousal Disorder_Controversies
- Apr 18
- 19 min read
Updated: 2 days ago
Background Information
Rao, N. (2026, April 18). Pathologizing Difference: A Critique of Sexual Interest, Arousal and Desire Disorders. N. Rao Publications. https://doi.org/10.5281/zenodo.19642402
Tiefer L., et al. (2003). New View of Women's Sexual Problems: A Teacher's Manual. FSD Alert. http://www.fsd-alert.org/media/pdfs/NVCteachingmanual.pdf
Tiefer L., et al. (2003). Female Sexual Dysfunction: A Case Study of Disease Mongering and Activist Resistance. PLoS Medicine. https://journals.plos.org/plosmedicine/article/file?id=10.1371/journal.pmed.0030178&type=printable
APA
In 2012, the American Psychological Association (APA) Council of Representatives apologized to BIPOC for their role in "promoting, perpetuating and failure to challenge Racism, Racial Discrimination and Human Hierarchy" in the Resolution (Auguste et.al., 2021).
APA rejects “hegemonic science”—that is, research focused on identifying and reinforcing supposed hierarchies of human value based on a White-default—and will continue to oppose it through culturally responsive training, ethical/equity-focused approaches, peer review, and publications (e.g., APA, 2021d).

A study analyzing psychology research conducted across a 2 year period (2019—2020), stated that while 90% (113/125) studies reported age, only 43% (54/125) reported race or ethnicity. Of those 54 studies, 94.6% participants identified themselves as White/Caucasian. In Non-genetic studies, White representation was 71.4%, Other/Non-white at 10.6%, African-American/Black/African at 7.6% and Hispanic/Latino at 5.3%. The remaining racial groups were <1%. Moreover, Asians/Asian-Americans were underrepresented in 78% of the articles, while Biracial/Multi-racial participants were underrepresented in 49% of the articles, and Black and White participants were underrepresented in 29% and 37% of the articles, respectively (Pedersen, S. L., et al., 2022).

Psychology research and practice is in its core, extremely W.E.I.R.D (Western, Educated, Industrial, Rich, Democratic). Across Psychology and Cognitive Sciences, over 90% of experimental participants come from the US (70%), Canada, and Western European Countries (Hruschkka, et al., 2018; Nielsen et al., 2017; Rad et al., 2018). In US alone, 94.6% of the participants have identified as White/Caucasian. Only about 5-8% of the participants in Psychology and Cognitive Sciences research, come from Non-American & Non-European countries, which make up the Global Population of 87%.

APA will encourage psychologists and trainees to consider the limitations of White Western-oriented clinical practice, and gain awareness of other healing approaches emanating from Indigenous and other non-Western and cultural traditions. APA will continue to learn and update new information on racism in diagnosis and clinical practice, and on the pursuit of equity, diversity, and inclusion in health service psychology, including psychological testing and assessment, while fostering practice based in culturally relevant evidence (American Psychological Association, 2021).
Just think about it. 87% of the Global Population only see a representation of 5-8%, while 13% of the Global Population see a representation of 90% in studies. This goes beyond just representation. As researchers predominantly, grew up in WEIRD societies, trained in WEIRD concepts and even in institutions, their assumptions, language, intuitions, reasoning, expectations, consciousness, everything was WEIRD.
Universalism is embedded in Western research and in the Mental Health Industrial Complex (MHIC). The equation remains the same. It begins with erasure of dispossessed voices, to theorize, quantify and medicalize/pathologize human conditions that are non-normative within the represented culture, aka, White-cis-het-male-able bodied-English speaking. Any behaviors or conditions that are non-normative now get diagnosed, coded and in Fanon's words, the people get "pacified" (Fanon F., 2004). Mental Health Oppression further oppresses those relegated to the margins with the same "DSM" that was originally built on a white-normative culture. It carries with it, the stain of White Supremacy, Ableism, Imperialism, Fatphobia, Transphobia, by reinforcing oppressive labels in order to get people to conform (Mullan, J., 2023).
While practitioners continue to practice in accordance to the violent history informing terms and conditions their license adheres to, very few challenge the system as a whole. It is bizarre that we continue to therapize, medicate and "heal" individuals while the system is allowed to go unchecked, continue its perpetration with no consequences. A Freirean perspective suggests that the current system often acts as an instrument of social control, treating systemic problems (racism, poverty, classism) as individual deficiencies, which "pathologizes the oppressed rather than the systems that oppress them" (Freire, P., 2018).

Mental Health Industrial Complex
The conversations about Health, Science, Medicine, Access, Safety, all have roots in Eugenics, Ableism, Desirability and Population Politics. And those conversations often lead us back to the colonizer's blueprint: the idea that many colonizers believed it was their birthright to invade a land, violently dispose of its people and knowledge bases, to impose one's own knowledge, to leave a legacy of dividing the residents into hierarchies based on color, class, caste, religion, sex, gender, sexuality, size and ability, and to continue to imperially impose violent social control by dehumanizing the population, destabilizing their governments, policing, infantilizing, displacing and imprisoning bodies, enslaving the people, assaulting and violating them, criminalizing their presence, pillaging their land and resources, destroying their balance with nature, desecrating their monuments.

The truth is that colonization, in its very essence, already appeared to be the great purveyor of psychiatric hospitals. (Fanon F., 2004)
The very nature of colonization is to create a dependency on systems by pacifying the colonized people. Fanon argued that the inherent violence, stress, and oppression of colonial rule "sows seeds of decay" that damage the mental health of the colonized, leading to a constant need for psychiatric institutions. With the reduction in diagnostic heterogeneity and neglection of context, DSM risks a reduction to Checklist Medicine, failing to address pathologization and medicalization of human differences in expression of behaviors and symptoms.
"We speak of burnout and exhaustion as the fault solely of the person experiencing it. Burnout is a form of late-stage capitalism."
For much of psychiatric history, the focus was on finding the root cause, until 1980 when psychiatric nosology moved away from being etiology-based to a descriptive, symptom-based correlational model. Often described as a checklist psychiatry, where diagnosis was made by meeting a specific criteria (which was WEIRD defined), mental health industry oversimplified complex human behaviors, leading to higher rates of over-diagnosis, diagnostic comorbidity and low validity. Now, it is important to differentiate between the American Psychological Association and the American Psychiatric Association, the latter of which is responsible for publishing the DSM. Despite the differences in professions, psychologists still use the DSM for diagnosis and billing. So let's talk about the Medical Industrial Complex that produced the manual of diagnostic criteria, the Diagnostic and Statistical Manual of Mental Disorders (DSM-5 & DSM-5-TR).
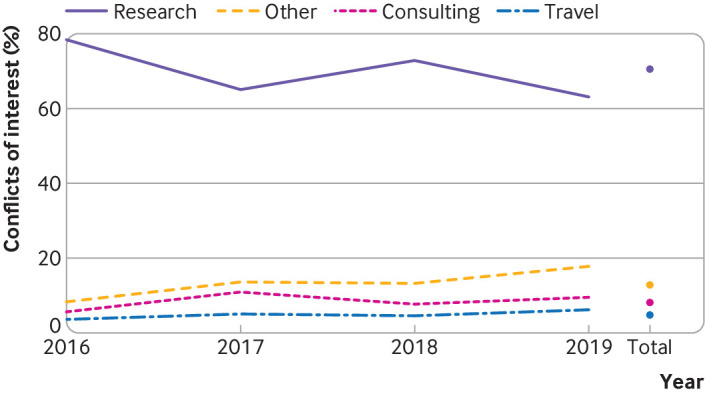
In 2024, a research group undertook the task to connect the dots between the Pharmaceutical Industry and the DSM-5 Panel, as well as its revisionist task force, DSM-5TR. 186 members of the panel and task force was identified, with 72 excluded from the study (these included 62 non-medical personnel and 14 outside US members). Of the 92 individuals who met the inclusion criteria, 55 had conflicts of interest. Of the 92 members (86 panel and 6 task force), 60% of them received financial compensation from pharmaceutical industries (the data is available in the slideshow below). Cumulatively, members were paid $14.2 million during the time period for constructing and revising the DSM (2016 — 2019). This creates a pro-industry bias influencing the diagnostic criteria the whole world relies on.
DSM-5 & DSM-5-TR
Over the past few years, DSM and its panelists have come under extreme scrutiny. It often operates by individualizing symptoms, rather than looking at the system as a whole. Ever DSM revision, further contributes to the increase in diagnoses, thereby pathologizing a wide-array of human behaviors, experiences and existences, that do not fit the normative white-cis-het-male bias. DSM-5 has also been criticized to have allowed the pharmaceutical industry to manipulate and influence revision processes, leading to an increased tendency to medicalize behavior and mood that are considered "inconsistent". These financial ties to the pharmaceutical industry, allows for an expedited pharma pipeline whereby the drug in question can be FDA approved with greater ease and profitability.
While pharmaceutical companies' goal is profit, MHIC supposedly prides itself in easing suffering. With over-medicalization and pathologizing human differences, the reality of the role played by MHIC however, tells a different story. The APA does make a clear statement regarding what is considered a mental disorder, and that is "a manifestation of a behavioral, psychological or biological dysfunction in the individual." It does make a distinction stating deviant behaviors, forms of oppression and inequity between people and society are not considered mental disorders. The language is inherently political, creating pathology and division (T. Smith, 2012). Diagnostic systems built on historically narrow populations can misinterpret or misclassify culturally different expressions of behaviors and symptoms, and when those systems are institutionalized, they can produce unequal and sometimes biased diagnostic outcomes.
Which begs us to ask the question—What is Normal? Who gains from creating definitions and continuums of what is considered Normal? Who loses their voice, their right to self-express and exist, pushed to the margins, dispossessed? Which culture's norms do we reference when talking about Normal? From whose lens are the margins for Normal being drawn? Western conception of normal deliberately excludes the BIPOC people.
For example, gender dysphoria was at one point a gender identity disorder, which until recently (2013) was not replaced by APA. Even after the edit, gender dysphoria puts the onus on the gender non-conforming person for failure to affiliate and adopt the gender binary. A person is only considered disabled in a society that was not built for them. Disability is caused by environmental and societal barriers rather than by an individual's impairment.
Medicalization, sociological term is defined by the transformation of concepts, authority and interventions from legal and religious to biomedical (Conrad & Schneider,1980). Medicalization is the process by which nonmedical problems become defined and treated as medical problems often requiring medical treatment.
The social model of disability emphasizes that inaccessible infrastructure and attitudes, not personal conditions, create limitations. Psychology and Psychiatry carry with them, the weight of questionable theories and human experimentations. Instead on fixing the system and focusing on making an inclusive environment, MHIC often plays the role of the perpetrator of pacification, enforcing the subjugation of its subaltern, the pacified subjects, so as to dictate a colonial, imperial and White-centric normative onto the population.
"We had no control over the fact that the psychiatric phenomena, the mental and behavioral disorders emerging from this war, have loomed so large among the perpetrators of pacification and the pacified population." (Fanon F., 2004)
And indeed, with an expedited entry into the DSM, many pathologized human conditions made their way through the pharmaceutical pipeline, bypassing the FDA regulatory bodies. Some would launch PR campaigns influencing FDA authorities, while some would bribe the authorities, many would release the drug/register for patents for its formulations in a foreign country, and utilize the paper trail to influence FDA's decision.
Since 1954, attention was called upon international psychiatrists who stated they had the “difficulty of curing a colonized subject correctly.” Essentially, having trouble enforcing a colonial ideal of normality, which was developed keeping the colonizer’s environment in mind, onto the
colonized. After Addyi, the "female Viagra" was released, it procured more patents and rights to distribute in other countries. Sprout Pharmaceuticals are now in negotiation with the Indian authorities to distribute Addyi. Moreover, when FSIAD was drafted into the DSM, the research terrain shifted to utilize the diagnoses (one built on only White data) to diagnose people from the Global South.
Female Sexual Interest and Arousal Disorder (FSIAD)
In May 1997, clinicians and researchers were invited to Cape Cod Hotel by 9 drug companies "to discuss the future and direction of clinical trials", and arrive at an agreement on what constitutes Female Sexual Dysfunction. In October 1998, at the 1st International Consensus Development Conference, Boston, a "closed session" was organized by the American Foundation for Urologic Disease, where participants were handpicked for their clinical expertise and their position as "thought leaders". They worked on the DSM-4 to produce definitions and classifications of Desire, Arousal, Orgasm and Pain Disorders. 18-19 of those authors had financial links to 22 companies.
Then came the 1999 JAMA article, which was even cited during the FDA proceedings. It stated that the total prevalence of sexual dysfunction was 43% among women aged 18-59. Both the authors of the paper had links to Pfizer. This article was later challenged as the critics found that if a woman answered yes to even one out of seven yes or no questions asked, she would be categorized as having a sexual dysfunction.

On the international stage, FSD was making moves. It was discussed at the 1st International Consultation on Erectile Dysfunction in Paris (1999). It was also decided that a second consultation would be necessary in June 2003, where an internationally accepted instrument for assessing sexual function would be adopted. This was sponsored by the World Health Organization (WHO), the International Society for Impotence Research (ISSIR, now the International Society for Sexual Medicine or ISSM), and the Société Internationale d'Urologie (SIU). Except for the WHO, the other 2 organizations are neck-deep in financial sponsorship/industry partnerships. ISSIR/ISSM in its comprehensive history, published in Sexual Medicine Reviews in 2021, mentions the establishment of an International Sponsors Board in 2003. The board was funded by 3 giants in the ED industry, Pfizer (Viagra), Lily-ICOS (Cialis) and Bayer (Levitra). This board lines up perfectly as the Second International Consultation in Paris in June 2003, was heavily sponsored by ISSIR/ISSM.
2000 & 2001 saw more conferences organized by Female Sexual Function Forum, each of these were funded by 22 drug companies, Pfizer being the key sponsor. 2002, the Female Sexual Dysfunction : Promises, Prescriptions & Profits conference was organized in San Francisco. 2002 also saw 15 drug sponsors at the annual meeting at the International Society for the Study of Women's Sexual Health, Vancouver. And that's how the Female Sexual Dysfunction was funded an elevated into the Mental Health Industrial Complex by many Drug Companies.

But that was from a Financial Liability stand-point. What about the language of the disorder itself? The Female Sexual Interest & Arousal Disorder has elements of Hyposexual Desire Disorder (HSDD), Female Sexual Arousal Disorder (FSAD) and Female Orgasmic Disorder (FOD). My critiques are only with reference to pathologizing Interest, Arousal, Orgasm and Desire, and not with Genito-Pelvic Pain as the latter has substantial, credible literature to support its diagnostic criteria. Fascination with women's "libido" has long been reified by pharmaceutical interests. In fact, the shift from HSDD to FSIAD added "unreceptiveness to partner's attempt to initiate sex", as if to say informed consent doesn't matter here and that all we need is to drug women and they will appear more suggestible. And since, this leads to pathologization and overmedicalization, at what point do we press pause and re-examine & re-interpret the "evidence", before this profit-driven diagnosis impacts not only America but also the rest of the world?
Addyi | Sprout Pharmaceuticals

Even the Score Campaign
After the 2nd FDA rejection, Sprout Pharmaceuticals hired Blue Engine Media to create a fake Feminist PR campaign to push the FDA into approving the drug. The campaign materialized 4 months before the FDA's patient-focused drug development meeting, and disappeared right after Addyi was approved. The FDA's Head of Office of Women's Health, Audrey Sheppard, was also flibanserin's biggest advocates: in 2013, Sheppard was organizing luncheons, conference calls and private meetings with feminist leaders (Block and Canner, 2016).
Moreover, Even The Score was never a feminist organization, it was a PR Campaign. It was yet another drug for men where their expectations of sex was catered to, all the while their partners were being drugged. The day after Addyi was approved, the campaign launched a 30 second video to thank the FDA. It features a man on the beach celebrating his new-found sexual appetite for his partner. He suddenly sits up and says, "Hey, did you guys hear the FDA just approved the first ever medication for women’s sexual desire? This gives a whole new meaning to my four-hour erection!", while the woman responds with, "I know what to do with it." From advertisements suggesting that female sexual dysfunction is also because of painful sex, indicating a woman teaching a man how to insert his "pointy side of the umbrella" into the sand, saying it needs to be gently put in and not jammed in, to feminist propaganda on the website stating 26 FDA approved drugs for men and not even 1 for women, Even The Score, went all in to promote the product from a feminist angle, a product with no credible research done to back its claim.
It’s known as Viagra for women – the little pink pill – a tiny tablet that can have a huge impact in the bedroom …. But you won’t be able to get it just yet, because the drug is locked in a heated battle for FDA approval, raising questions about why there are so many drugs out there for sexual dysfunction for men. And zero for women. (Schiavocampo et al., 2014)
Much of Even the Score's messaging was that the pill would save marriages. Not surprisingly, local women (including Amanda Parrish, the mother of 4) and advocates for the pill that was covered in the marketing, were all paid for by a Sprout-sponsored meeting-organizing group, Veritas. Oct 14, 2014, ISSWSH, one of the 26 organizations sponsoring the Even the Score, wrote to their member physicians to ask their FSD patients to attend FDA-patient focused meeting.
"If your patients are passionate about this issue and are able to attend the FDA patient panel, funding is now available (money is from multiple sources) to help support travel and hotel costs for patients to attend the meeting. Patients can request a travel grant by writing to sue@veritasmeetingsolutions.com." (ISSWSH Email, 2014)
FDA Patient-Focused Meetings (2013-2017)
FSD was added to the ranks of HIV, Parkinson's and Sickle-cell, a drug category with "unmet drug needs" (Cacchioni, 2014). 30 women were called in to narrate their experience. After the meeting, Even The Score uploaded pictures of these women, alongside some of the husbands during the briefing session which was prior to the FDA meeting (they were seated in the bus, posing in front of FDA HQ, many wearing Even The Score buttons and scarves).
Its rather unfortunate that at the federal level, an organization that was uncorruptable, was able to be swayed. Even the suggestion of talk therapy as a modality was dismissed because the women felt that "No amount of talk therapy is going to fix [this problem]". They assumed that the drug was effective, that it has a reliable mechanism. But this drug has only been tested in vivo and lacks the human FMRI studies to back its claims. Moreover, the participants were keen on rejecting that the "problem was all in their heads" which is ironic as the drug they were advocating for supposedly works by releasing certain neurotransmitters.
Moreover, FDA proceedings demand a range of perspectives to be gathered. Then why is it that five out of eight women selected as speakers by the FDA, introduced themselves as such:
My name is Vicki … . Veritas has tak[en] care of my travel expenses through grants from Sprout Pharmaceuticals, Even the Score, and the Institute for Sexual Medicine (FDA, 2014: 47).
My name is Beverly. And Veritas arranged my travel here today (p. 51).
First of all I’d like … to thank Veritas for our travel arrangements …. My name is Judith (p. 127).
Hi, I’m Katherine. I need to thank Veritas Meetings who was given [a] grant by Sprout among others for covering my basic travel expenses and making it possible for me to be here today (p. 132).
[My name is Barbara]. I also want to thank Veritas for supplying the grant so that I could be here to speak to all of you (p. 135).
To further note here, is that the Sexual Satisfactory Event (SSE) was often described as the "husband's satisfaction", that the woman's satisfaction in sex depended on how satisfied the husband was. Here are some excerpts for you to peruse.
I would say something that would end up being pleasurable would be the fact that when it ends your husband doesn’t automatically think it was out of obligation. (FDA, 2014: 118) It is a success if he is having a good time … I might not even want to have sex but if he wants sex then and I give it to him then, yes, I was a good wife today. (p. 116) [With the use of testosterone] I can have multiple orgasms during a sexual encounter with my husband which makes him really happy. (p. 153)
I am still at a loss of words on how Even The Score was able to pull this as a "Feminist Stunt" when the pill was being marketed and used by women to obtain a placebo effect that would make their husbands enjoy having sex again. Where does it actually deal with women getting aroused or women having an interest or women enjoying? "Male pleasure was central to women's sexual activity" (2018). The pill was marketed so as to manufacture compliance on the women's part.
FDA’s questions all row in the same direction. No one asks if women who don’t reach orgasm with their male partners do reach orgasm when they masturbate – or if they are aroused while reading erotica. No one asks if they really expected their libido in their 40’s to match their libido in the 20’s – or their desire for their long-time husbands to match their desire for their newly wed ones (Klusmann, 2002).
Two days after Addyi was approved, it was sold for $1 million to Valeant Pharmaceuticals. It was then reacquired by Sprout citing "severe mismanagement". Valeant sold to Sprout shareholders, for an exchange of 6% royalty on Addyi's sales after 18 months. Sprout Pharmaceuticals and their founders have had a history of misleading their customers. Earlier with the Testosterone patch and this time with Addyi. FDA has issued several warnings to regulate these Addyi marketing stating they should not appear without their boxed warnings.
So far, despite the lack of inclusive studies, Addyi (flibanserin) has been marketed outside of the US & Canada to Egypt. To further complicate matters, Addyi was even investigated by the Congress in October 2015 over predatory drug practices. The company was accused of using Philidor Rx Services, a distributor to jack up the prices. After the investigation the company had to cut ties with its distributor. But why jack up the prices if over 50% women "have FSIAD"? Why increase the price if it is a good drug that is meant to help women? You see, insurance companies won't cover Addyi and its not a sexist explanation, its that the drug lacks efficacy.
If the drug lacks efficacy then why is it in the market? Who is its consumers? Who is more likely to get diagnosed with FSIAD? Upper-class, heterosexual, married White women. Not Queer, not Black and not Single women. So why do women on Addyi report it works if the efficacy is low? Well it has a lot to do with Manufacturing Consent/Interest/Arousal/Desire as opposed to increasing them. Recall women's needs from the FDA session — it was to satisfy their husband.
Mechanism of Action:
National Center for Biotechnology Information
Serotonin (5-hydroxytryptamine (5-HT)) is a well-studied neurotransmitter and hormone known for regulating mood and various peripheral physiological functions. Serotonin influences various physiological functions, including sexual desire. The positive regulation of libido is facilitated by the presence of dopamine (DA) in the synapse and other microcircuits, where the actions of estrogen, norepinephrine (NE), and testosterone occur. The negative regulation of libido occurs through microcircuits at sites where serotonin and prolactin exert their actions. This suggests that an imbalance in these microcircuits may contribute to the development of HSDD, indicating reduced levels of DA and NE and increased amounts of 5-HT.
Flibanserin selectively targets central serotonin postsynaptic receptors, exhibiting both agonistic and antagonistic effects on 5-HT1A and 5-HT2A, respectively. Flibanserin's activity on these specific serotonin receptors distinguishes it from other drugs targeting serotonin receptors. The activity of flibanserin on 5-HT1A and 5-HT2A receptors leads to elevated NE and DA levels and reduced serotonin levels within the prefrontal cortex intracellularly. Alongside its targeted effects on postsynaptic serotonin receptors, flibanserin exhibits limited antagonistic activity on 5-HT2B, 5-HT2C, and DA D4 receptors.Flibanserin is not associated with addiction or misuse potential, as it does not alter dopaminergic neurotransmission in the nucleus accumbens.
Absorption: In healthy premenopausal women, the oral administration of a 100 mg dose of flibanserin results in a time to reach a maximum plasma concentration of approximately 0.75 hours. The absolute bioavailability of flibanserin after oral dosing is 33%.
Distribution: Flibanserin binds primarily to albumin, exhibiting a plasma protein binding of approximately 98%.
Metabolism: Flibanserin undergoes primary metabolism through the cytochrome P450 enzymes CYP3A4 and CYP2C19, with minimal contributions to its metabolism made from CYP1A2, CYP2B6, CYP2C8, CYP2C9, and CYP2D6 based on in vitro and in vivo data. Flibanserin undergoes extensive metabolism, resulting in at least 35 metabolites, most of which are present in low concentrations in plasma. Among these, only 2 metabolites—6,21-dihydroxy-flibanserin-6,21-disulfate and 6-hydroxy-flibanserin-6-sulfate—exhibit plasma concentrations comparable to flibanserin. Notably, both of these metabolites are inactive.
Elimination: After a single oral solution administration of 50 mg 14C-radiolabeled flibanserin, 44% of the total 14C-flibanserin–related radioactivity is excreted in urine, whereas 51% is eliminated in feces. The average terminal half-life of flibanserin is approximately 11 hours.
Pharmacogenetics: Clinicians should be mindful of pharmacogenetic variations to mitigate the risk of adverse effects related to flibanserin, particularly in patients identified as poor CYP2C19 metabolizers. Individuals classified as poor CYP2C19 metabolizers may experience elevated levels of flibanserin, heightening the risk of adverse events such as hypotension, syncope, and CNS depression. This subgroup constitutes 2% to 5% of Caucasians and Africans and 2% to 15% of Asians.
Hepatic impairment: Mild hepatic impairment (Child-Pugh score of 5 to 6 points) showed that single 50 mg oral doses of flibanserin led to increased systemic exposure and a longer half-life than those with normal hepatic function. Due to the limited sample size in moderate hepatic impairment (Child-Pugh score of 8 to 9 points), conclusive statements regarding the impact of flibanserin exposure cannot be asserted. Notably, flibanserin is contraindicated for hepatic impairment.
Renal impairment: The pharmacokinetic data in the labeling indicates a slight increase in the area under the curve (AUC) by 1.1-fold in mild-to-moderate renal impairment and a 1.2-fold increase in AUC for severe renal impairment compared to healthy individuals. In addition, the data suggest that patients with renal impairment may not require dosage adjustments, as the observed alteration in AUC is minimal. The FDA-approved labeling does not specify any dosage adjustments for renal impairment.
Pregnancy considerations: An ongoing flibanserin pregnancy registry study assesses pregnancy outcomes in women exposed to flibanserin at any stage of pregnancy. To date, no studies on pregnant women have conclusively identified any drug-associated risks. However, studies based on animals have indicated certain adverse effects on reproduction and development, including diminished fetal weight, structural anomalies, and increased fetal loss, observed at exposures significantly exceeding the maximum recommended human dosage. Notably, although flibanserin is not recommended during pregnancy, it is also not contraindicated in this population. https://www.fda.gov/science-research/womens-health-research/list-pregnancy-exposure-registries https://addyipregnancy.com
Breastfeeding considerations: Although flibanserin has been detected in rat milk, its presence in human milk and its impact on the breastfed infant and milk production remains unknown. Due to the potential for adverse reactions, including sedation in a breastfed infant, breastfeeding is not recommended during flibanserin treatment.
Pediatric patients: Flibanserin is not approved by the FDA for use in the pediatric population.
Older patients: Flibanserin lacks FDA approval for use in older individuals, as its safety and efficacy in this age group have not been confirmed.
American Psychological Association. (2021, October 29). Apology to people of color for APA's role in promoting, perpetuating, and failing to challenge racism, racial discrimination, and human hierarchy in U.S. https://www.apa.org/about/policy/racism-apology
Borsini, F., Evans, K., Jason, K., Rohde, F., Alexander, B. and Pollentier, S. (2002), Pharmacology of Flibanserin. CNS Drug Reviews, 8: 117-142. https://doi.org/10.1111/j.1527-3458.2002.tb00219.x
Conrad, P., & Schneider, J. W. (1992). Deviance and medicalization: From badness to sickness (Exp. ed.). Temple University Press.
Email from the ISSWSH Executive Office to ISSWSH members, forwarded by Leonore Tiefer to the New View listserv on 6 October 2014
Fanon, F. (2004). The wretched of the earth (R. Philcox, Trans.). Grove Press
Freire, P. (2018). Pedagogy of the Oppressed (50th Anniversary ed.). Bloomsbury Academic
Hruschka, D. J., Medin, D. L., Rogoff, B., & Henrich, J. (2018). Pressing questions in the study of psychological and behavioral diversity. Proceedings of the National Academy of Sciences, 115(45), 11366–11368. https://doi.org/10.1073/pnas.1814733115
Jutel, A. (2010). Framing disease: The example of female hypoactive sexual desire disorder. Social Science & Medicine, 70(7), 1084–1090. https://doi.org/10.1016/j.socscimed.2009.11.040
Mecham, S., & Rickard, A.J. (2018). The Usage of Flibanserin to Treat Women ’ s Hypoactive Sexual Desire Disorder in the U.S.
Moynihan R. (2003). The making of a disease: female sexual dysfunction. BMJ (Clinical research ed.), 326(7379), 45–47. https://doi.org/10.1136/bmj.326.7379.45
Mullan, J. (2023). Decolonizing therapy: Oppression, historical trauma, and politicizing your practice. W.W. Norton & Company.
National Center for Biotechnology Information (2026). PubChem Compound Summary for CID 6918248, Flibanserin. Retrieved April 18, 2026 from https://pubchem.ncbi.nlm.nih.gov/compound/Flibanserin.
Nielsen, M., Haun, D., Kärtner, J., Legare, C. H., Kartner, J., & Legare, C. H. (2017). The persistent sampling bias in developmental psychology: A call to action. Journal of Experimental Child Psychology, 162, 31–38. https://doi.org/10.1016/j.jecp.2017.04.017
Pedersen, S. L., Lindstrom, R., Powe, P. M., Louie, K., & Escobar-Viera, C. (2022). Lack of Representation in Psychiatric Research: A Data-Driven Example From Scientific Articles Published in 2019 and 2020 in the American Journal of Psychiatry. The American journal of psychiatry, 179(5), 388–392. https://doi.org/10.1176/appi.ajp.21070758
Pierrelus C, Patel P, Carlson K. Flibanserin. [Updated 2023 Nov 10]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2026 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK589649/
Rad, M. S., Martingano, A. J., & Ginges, J. (2018). Toward a psychology of Homo sapiens: Making psychological science more representative of the human population. Proceedings of the National Academy of Sciences, 115(45), 11401–11405. https://doi.org/10.1073/pnas.1721165115 Segal, J. Z. (2018). Sex, drugs, and rhetoric: The case of flibanserin for ‘female sexual dysfunction.’ Social Studies of Science, 48(4), 459–482. https://doi.org/10.1177/0306312718778802
Spurgas, A. K. (2013). Interest, Arousal, and Shifting Diagnoses of Female Sexual Dysfunction, or: How Women Learn About Desire. Studies in Gender and Sexuality, 14(3), 187–205. https://doi.org/10.1080/15240657.2013.818854
Sungur, M. Z., & Gunduz, A. (2013). Critiques and Challenges to Old and Recently Proposed American Psychiatric Association’s Website DSM 5 Diagnostic Criteria for Sexual Dysfunctions. Klinik Psikofarmakoloji Bülteni-Bulletin of Clinical Psychopharmacology, 23(1), 113–128. https://doi.org/10.5455/bcp.20130416063859









Comments